The Challenge of Childhood Cancer
The causal picture is complicated. Genetics offers a way forward.
This article is part of a series on hereditary cancer syndromes and cancer genetics called Cancer Genomes. If new to the series, please go to my post “Introducing Cancer Genomes” for an explainer.
In November 1983, the broadcast ‘Windscale—The Nuclear Laundry’ aired on British TV, alleging a significant rise in cancer cases near the Sellafield nuclear facility. The program specifically highlighted an alarming increase in leukemia cases, reporting an alleged tenfold surge among children under 10 in a village called Seascale. This sparked profound concern among the local populace. It wasn’t long before the heightened cancer rates in children were linked - in the minds of Seascale residents - to the plant’s emissions. This was quick connection was likely fomented by a history of accidental leaks, the most infamous being the Windscale Fire of 1957.
In rapid response, the British government established a specialized Independent Advisory Group, known as the ‘Black Committee,’ to examine the allegations. After a quick yet thorough study, the Black Committee published a report that acknowledged the high leukemia rates among individuals under 25 years old, but it concluded that the radiation levels experienced by the community were insufficient to explain the case burden. Consequently, the committee advised additional research into the notable rise in cases and the radiation exposure of the local residents.
In November 1985, the British government set up the Committee On Medical Aspects of Radiation in the Environment (COMARE) to continue to monitor and study childhood cancer clusters around nuclear installations. After more than three decades of research, COMARE in concert with the Childhood Cancer Research Group (CCRG) has yet to demonstrate that radiation from these plants is contributing to the childhood cancer cases. Subsequently, it is possible these “clusters” are the byproduct of a sort of gerrymandering (i.e. case load inflation relative to the true background population due to arbitrary boundaries), ascertainment bias (i.e. undue focus on areas near nuclear facilities), and publication bias (i.e. ignoring areas around nuclear facilities without purported clusters of childhood cancer).
I don’t raise this anecdote to argue that environmental radiation is never a contributing factor to childhood cancer outcomes nor to argue that clusters of childhood cancer have never occurred. I have highlighted the potentially false mystery of Sellafield to illustrate the complex issues at play in childhood cancer etiology. The Sellafield dataset could have been made incalculably more valuable with better methodological approaches and with 21st century genomic techniques. One hopes that in future cases of suspected environmental exposures that government and research institutions can step up and definitively resolves such questions.1
Describing Childhood Cancer
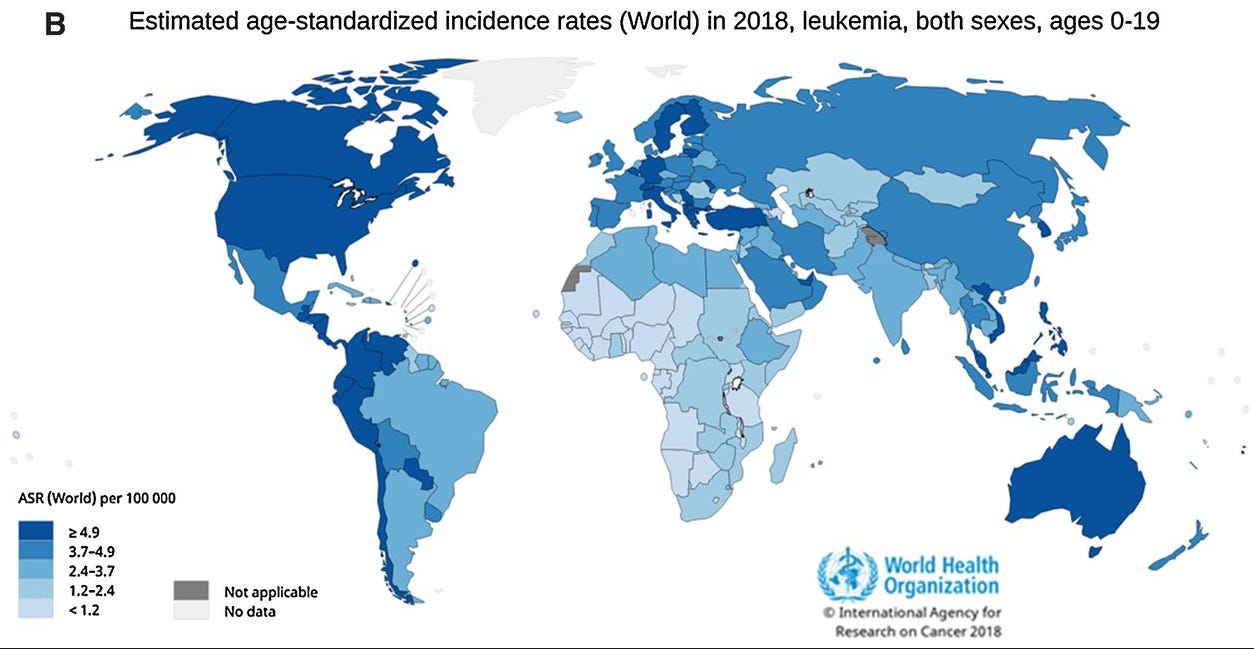
In the prestigious Lancet Oncology, Dr. Nickhill Bhakta and colleagues estimated the annual incidence of pediatric cancer (0-19) to be 300,000. Their estimate was derived from the Global Cancer Observatory (GCO) platform. Interestingly, they also observed that incidence of childhood cancer is highest in North America, parts of South and Central America, Europe, and Australia. In these often higher income places, the age-standardized incidence rate (ASR) was assessed at or above 15.4 per 100,000 person-years for those 0–19 years of age.
There are important limitations to this estimate. First, the coding of the data depends on diagnostic codes that are not a perfect match to natural childhood cancer groupings. Second, there is good reason to suspect some inaccuracy in the reported data from middle- and low-income countries. This is due to pathology of variable quality, misdiagnosis, and ascertainment failures. Subsequently, it is likely the empirical estimate is artificially deflated. A modeling study published alongside the empirical estimate found the annual incidence in a smaller age range (0-14) to be nearly 100,000 case higher.
Unfortunately, the biases in the data largely preclude tantalizing speculation about the origins of regional differences. However, these data do confirm that leukemias are the most common malignancy in children regardless of region. Plus, there are some regional differences that do seem to be explained by environmental factors. For instance, unmanaged HIV infection rates likely explain the much higher rates of Kaposi sarcoma in Sub-Saharan Africa and Burkitt lymphoma in North Africa and Sub-Saharan Africa.
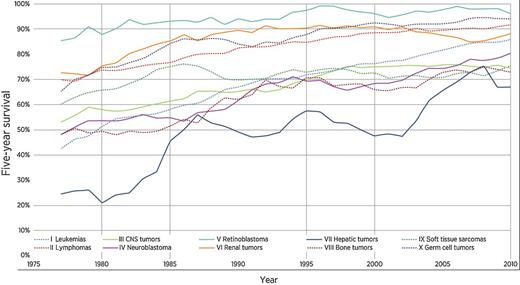
Outcomes for childhood cancers trend with country income. In the early 1990s through 2000s, the five-year survival rates were approximately 80%, 55%, and 40% for high-, middle-, and low-income countries, respectively. Leukemia and lymphomas had the best outcomes by cancer type. Today, the U.S. has roughly 1,200 annual deaths in children 0-15 from cancer. Despite generally low incidence (183 per million in the U.S.), pediatric cancers are collectively the number one cause of disease-related death in children living in high-income countries. Fortunately, since the 1960s, survival rates have steadily improved, increasing almost three-fold by the ends of the 2000s (28% —> >80%).
Before diving into the complex mix of causal factors, there are some demographic statistics of note. For the most common pediatric cancer, acute lymphoblastic leukemia (ALL), there is a distinct peak in occurrence between 2-5 years of age. Bone and soft-tissue sarcomas peak in middle adolescence, which also sees increasing incidence of lymphoma and other solid tumors. The age-specific trends contrast with the risk profile for central nervous system (CNS) tumors, which holds steady throughout youth. Interestingly, males have a higher risk for most childhood cancers. This trend only reverses (and only slightly) for Wilms tumor, extra-gonadal germ cell tumors, thyroid carcinoma, and melanoma. In the U.S. cancer rates by race/ethnicity show some differences. For instance, the rate of childhood cancer is 25%–50% lower in black children compared with white children, while the rate of ALL is 20% higher among Hispanic children compared with white children.
The Causes of Childhood Cancer
From my perspective, childhood cancer presents a likely case where genetic changes, whether inherited or sporadic, are contributing in outsized fashion to pathogenesis.2 This likely contrasts with the intuitions of the public. Hollywood depictions like Erin Brockovich (2000) or Dark Waters (2019) capture anxious imaginations. These stories offer simple explanations and solutions and thus get assigned an disproportional share of blame. The tragic reality is more complex and certainly beyond simple fixes. Inherited variation, stochastic genetic/non-genetic changes, and environmental exposures and factors are all important parts of the etiological picture. I simply think the evidence and available paradigms for study suggest we should start with genes.
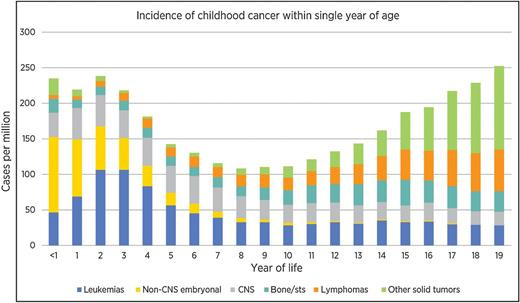
A quick look at the incidence pattern of pediatric cancers (0-19) previews the complexity at play. The incidence pattern takes a parabolic shape. The first crest is in very early life (0-4) followed by a lull in middle childhood through early adolescence (5-14), then it picks up again, cresting at the end of late adolescence (15-19). Throughout there are also changes in the proportion of cancer subtypes. Two decades of life is a small window for exposures relative to the eight decades of a typical adult life. Even in adults, environmental insults tend to be marginal or they contribute in quite specific ways to risk and pathogenesis.3 We tend to be quite familiar with these examples, especially given the public efforts dedicated to reducing carcinogenic exposures, e.g. smoking cessation campaigns. At the very least, the early life crest of incidence suggests tumorigenesis has likely kicked off in utero. This implicates inherited or early genetic changes.
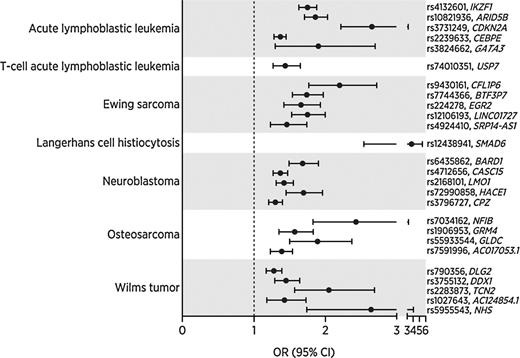
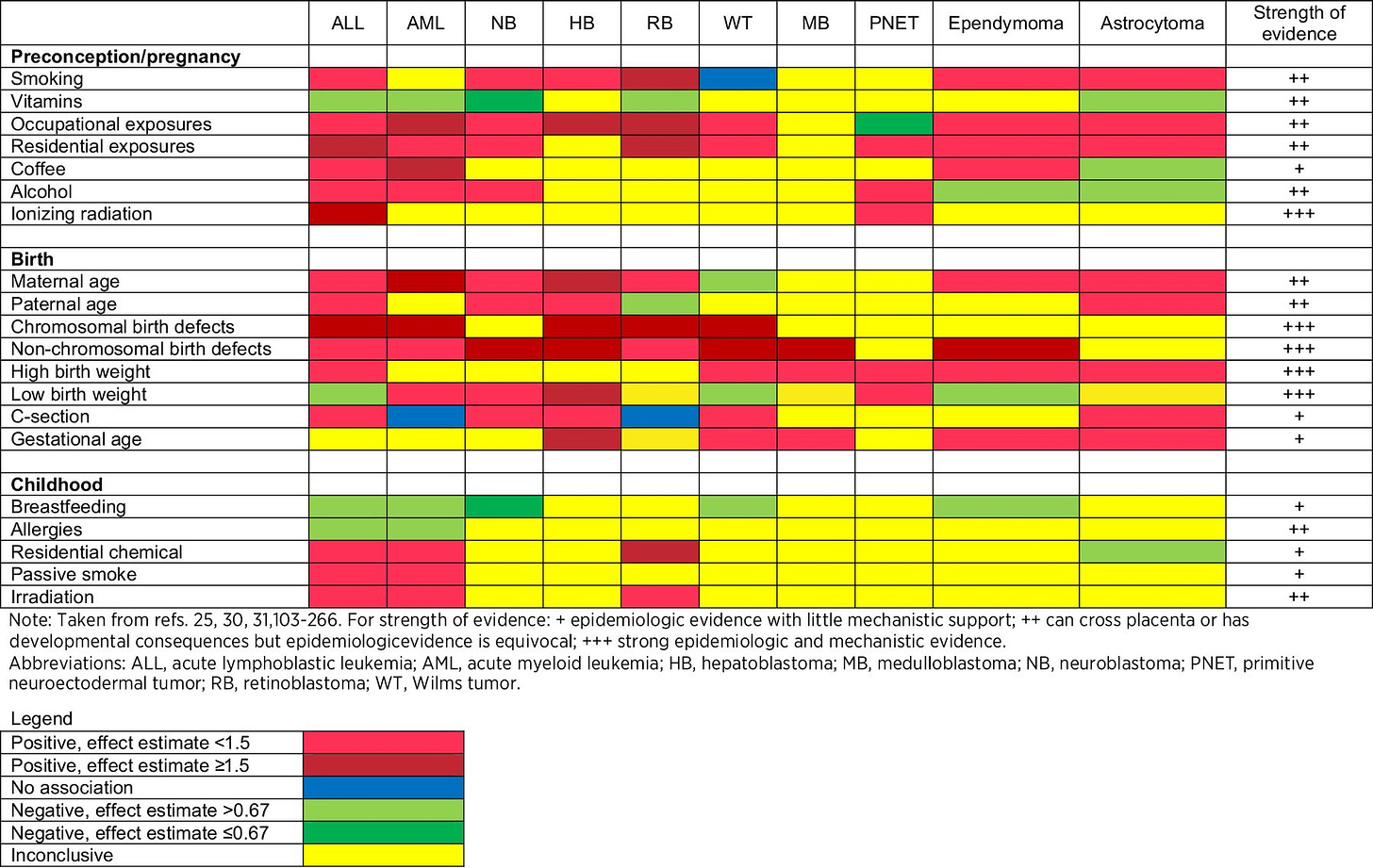
Nonetheless, compared to adult cancers, the etiology of pediatric cancers remains more ambiguous. This is due in part to the rarity of cases. This limits samples sizes and the type of research that can be conducted. It is frustrating challenge. Despite these challenges, some case-controls studies have provided tentative insight into risk factors, which are summarized in the chart below. In some respects, I am loathe to even show this chart because of its potential to mislead. The evidence for many of these risk factors is underwhelming and proportional to disease incidence. The authors that created the chart admirably tried to contextualize these issues, but I think it is still likely they are overstating the effects and strength of evidence in most cases.
Genetic Risk Factors (Rare Variation)
One of the clear risk factors is inherited variation in genes. Although explaining only a minority of cases, chromosomal abnormalities, structural variants, and pathogenic germline single nucleotide variants (SNVs) confer a sharply increased risk of cancer. In many pediatric cancer types, cohort studies using next-generation sequencing (NGS) have produced precise estimates of the prevalence of germline pathogenic mutation. For example, half or more of adrenocortical carcinoma cases carry such mutations. This is a high rate of germline mutation that contrasts starkly with the consensus rate of germline mutation observed in adult cancers, 5%–10%.

A 2015 study of germline mutation prevalence in 60 cancer risk genes across roughly 1100 pediatric cancers demonstrated that many subtypes showed mutation prevalences greater than 10%: osteosarcoma, retinoblastoma, adrenocortical carcinoma, and hypodiploid ALL. This same study also found evidence of mosaicism suggesting post-zygotic de novo events and admitted that it likely underestimated the relevant germline findings.4 Other sequencing studies have replicated these finding. Research on the subject consistently yields rare, highly penetrant variants and moderately rare, moderately penetrant variants likely responsible for pediatric cancer risk. I hope to see a systematic effort to screen for pediatric cancer predisposition in the near future despite the challenges this presents. It is abundantly clear that genetic variation is the best described, understood, and manageable risk factor for childhood cancer.

Genetic Risk Factors (Common Variation)
A 2020 cohort study of roughly 750 patients with pediatric cancer with ~16,000 mapped relations to the third degree demonstrated an increased standard incidence ratios (SIR) of cancer that was robust to removal of rate pathogenic mutations. This study is a neat epidemiological illustration of shared cancer risk from likely common genetic sources in families with pediatric cancer cases. Similar conclusion have also been supported by genome-wide association studies (GWAS) despite early failures.
Currently, GWAS has produced mature genetic architectures with multi-ancestry replication5 and ancestry-specific risk loci6 for ALL and neuroblastoma. There have been many other published GWA studies for various pediatric cancers: Ewing sarcoma, Wilms tumor, and osteosarcoma. Recent work has also shown that pediatric cancer associates with trait-related variation like height and telomere length. These trait association are fascinating to contemplate and suggests interesting research possibilities for those that can assemble large enough samples for similar work. Additionally, there remains a great deal of work to do when it comes to assessing the functional effects of the GWAS hits for pediatric cancer.
The Future of Pediatric Cancers
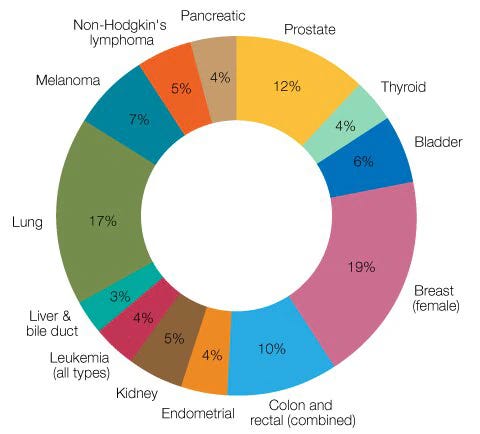
Pediatric cancer are distinct from their adult counterpart. Genetic risk factors likely figure more prominently, and the distribution of cancer types are distinct. Solid tumors are overwhelmingly dominant among adults, while blood cancers are significantly enriched among children. The breakdown is roughly 90% solid and 10% blood in adults, while it’s closer to 50:50 in pediatric individuals. These differences highlight the many incompletely answered and unanswered questions. Answering these questions should be a priority as increasingly we have the technology and resources to do so.

Recent advances in -omic approaches and technologies have enabled deeper molecular subtyping of pediatric cancers. ALL, glioma, medulloblastoma, and rhabdomyosarcoma have all seen recent subdivisions based on molecular features. We can take this deep phenotyping approach further and marry it with other methods. Precision medicine paradigms that have shown success in adult cancer and modern molecular epidemiology can both contribute significantly to pediatric cancer care. When we take this research further and integrate the findings of different approaches, we will improve our understanding of the causes of this tragic disease and identify new ways to stop it.
For more background on ionising radiation and childhood cancer, check out this 2018 review from Gerald M. Kendall and colleagues.
This was recognized all the way back in 1976 by a legend of cancer biology, Dr. Alfred Knudson. Knudson argued “the main effort against childhood cancer must be that of early diagnosis and treatment” because of the likely contribution of difficult to modify risk factors like genetic mutation, inherited and sporadic. However, he did suspect a possible viral contribution to pediatric blood cancers, which has not been demonstrated.
For adults, the most important explanatory risk factor is the aging process itself. Aging is thought to mediate cancer risk via two primary mechanisms: somatic mutation burden and immune senescence.
The likelihood of underestimation stems from a number of factors, including a limited gene list, limited sequencing depth, variants of unknown significance, and evaluated variant type.






Interestingly, I would highlight that in medical school, we are taught to really associate the leukemias, including pediatric leukemias, with genetic underpinnings, perhaps more so than any other cancer (expect HBOC or Lynch Syndrome probably). So at least there is some traction in this capacity!
Great synopsis of the field!